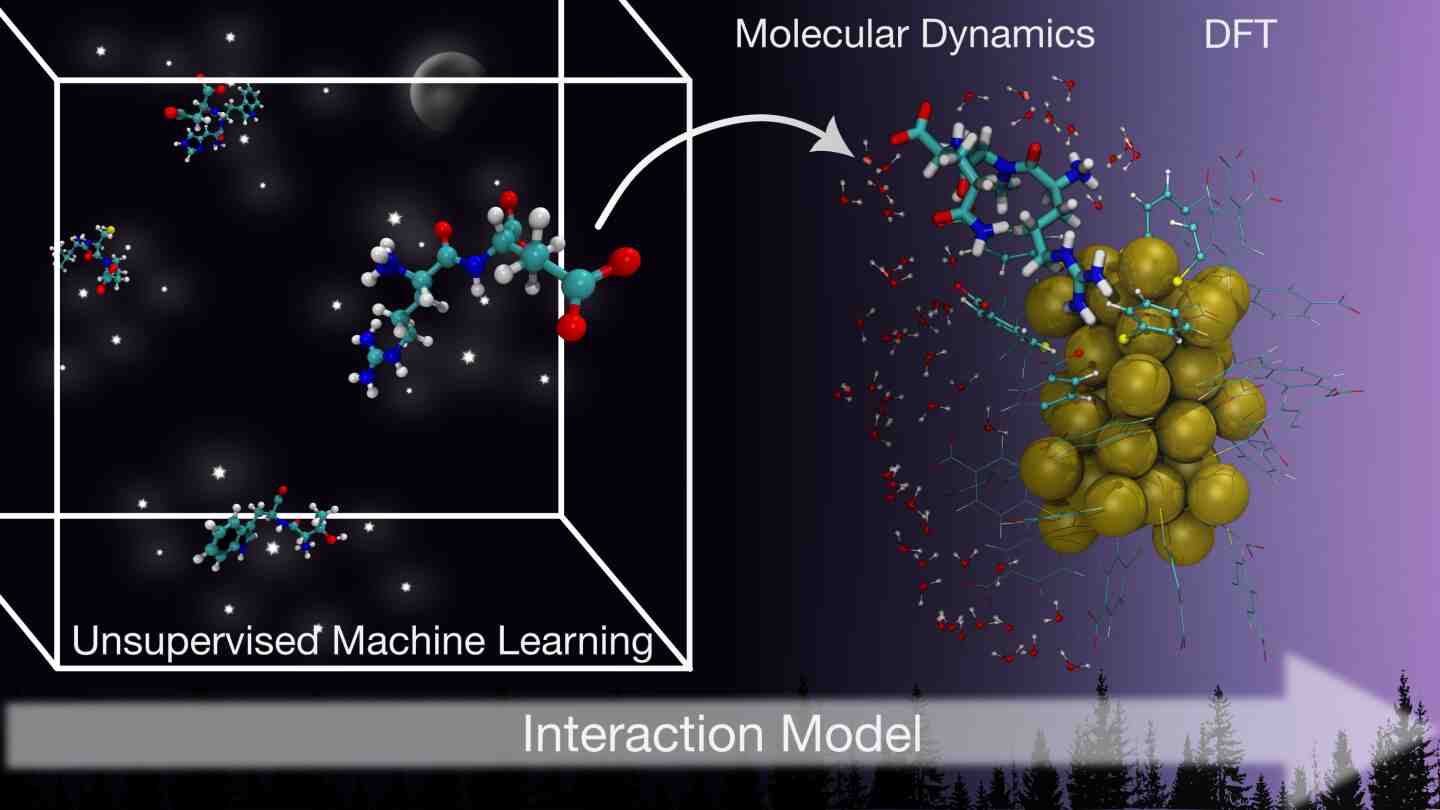
In a bold stride forward for computational nanoscience and biomedical innovation, researchers at the University of Jyväskylä’s Nanoscience Center in Finland have unveiled a groundbreaking machine-learning model that predicts how proteins bind to gold nanoclusters, a pivotal challenge in designing next-generation nanomaterials for bioimaging, biosensing, and targeted drug delivery. The work exemplifies how supercomputing, the very heart of high-performance computing, is accelerating discovery in fields that once lay beyond our computational reach.
At the core of this achievement is a novel clustering-based machine-learning framework that uncovers the chemical rules governing interactions between biomolecules and ligand-stabilized gold nanoclusters. Predicting protein adsorption at this level of detail has long stymied researchers due to the sheer complexity inherent in nanoscale interfaces. Traditional computational methods, even on powerful desktops, can require prohibitively long times and often lack the generalizability necessary to guide design across diverse proteins.
Here’s where supercomputing comes in. The team harnessed the LUMI supercomputer to perform atomistic simulations at an unprecedented scale and fidelity. These simulations provided the rich, high-resolution data necessary to train and validate the machine-learning model, a task virtually impossible without supercomputing resources capable of executing massive parallel computations with blistering performance.
Supercomputing enables scientists to tackle problems that are too large, too complex, or too data-intensive for conventional computing systems. By integrating hundreds or thousands of compute nodes working in concert, supercomputers like LUMI can complete simulations and data-driven training tasks orders of magnitude faster than standard hardware, dramatically shortening the cycle from hypothesis to discovery.
This synergy between machine learning and supercomputing yields not just faster computation, but also expands insight. The Jyväskylä model determines which amino acids are more or less likely to bind to gold nanoclusters and pinpoints chemical groups responsible for these interactions, a roadmap for rational design of nanomaterials with tailored properties. Importantly, the framework’s general and scalable design means it can be extended beyond a single peptide system to broadly inform how proteins interact with nanomaterials.
The implications are profound. With supercomputing-driven machine learning at their disposal, researchers can rapidly screen thousands of protein candidates and optimize nanomaterials for specific biomedical applications, from enhancing contrast in imaging to improving the specificity of drug delivery vehicles. What once required months or years of trial and error can now proceed at the speed of computation.
For the supercomputing community, this research highlights a powerful truth: the next wave of scientific breakthroughs will increasingly emerge where advanced algorithms meet extreme computing power. As the global high-performance computing ecosystem continues to evolve, with ever-faster machines and more sophisticated AI integrations, the frontier of what’s computationally possible will only expand.
In the words of the study’s lead researchers, this is not merely a model for a single system; it is a foundation for a new paradigm in computational nanoscience, propelled by the unparalleled capabilities of supercomputing.


 How to resolve AdBlock issue?
How to resolve AdBlock issue?